|
Copyright R. Zamenhof, 2013.
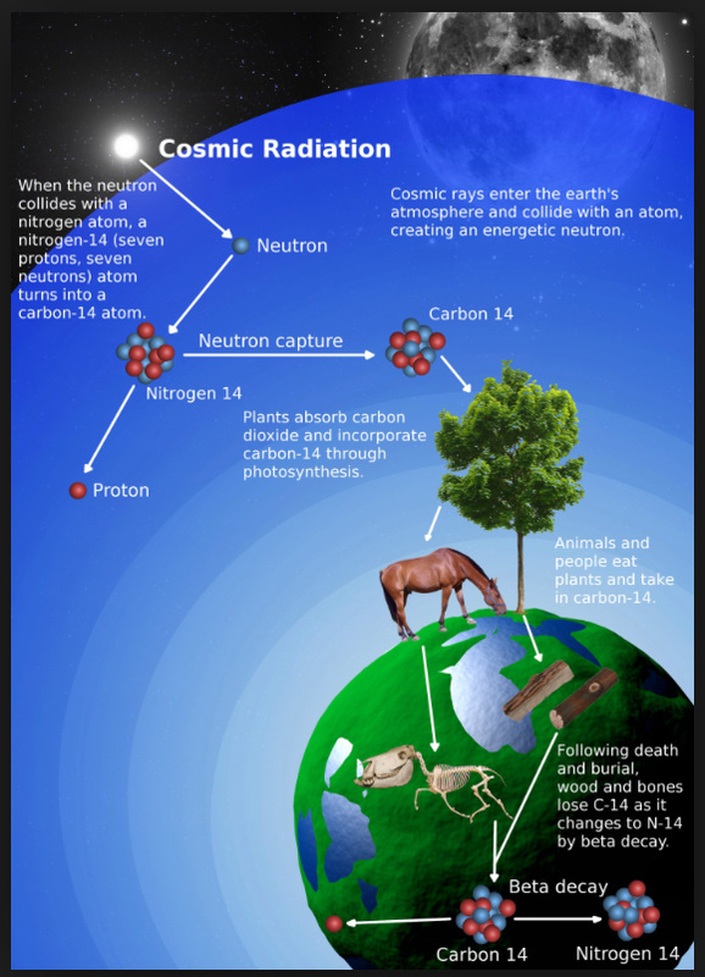 Diagrammatic depiction of the principles behind carbon dating. Carbon-14 (radioactive, with half-life of 5,730 years) is created in the atmosphere by nuclear interaction of cosmic rays and atmospheric nitrogen. Plants absorb the carbon-14 via carbon dioxide and people and animals absorb carbon-14 by consuming the plants. An equilibrium between carbon-14 in the atmosphere and in living organisms makes the carbon-14 / carbon-12 (non-radioactive) ratio remain constant. But when a living organism dies, no more carbon-14 is absorbed, so as the carbon-14 decays, the carbon-14 / carbon-12 ratio decreases with time. Measuring this ratio precisely provides the age of the organism at death Introduction
The general technique of radiometric dating was first published in 1907 by Bertram Boltwood, and is now the principal source of information about the absolute age of rocks and other geological entities. It can be used to date a wide range of natural and man-made materials including the age of the Earth itself. Carbon Dating of Organic Materials A specific subgroup of radiometric dating is called Carbon Dating, and is generally used to date plant and animal remains. Carbon dating was invented by Willard Libby in the late 1940s, and soon after became a standard tool for archaeologists and anthropologists. The most prevalent radionuclide found in the tissues of living plants and animals is carbon-14 (*C-14) (the asterisk is there as a reminder that the carbon isotope is radioactive). *C-14 is produced by the interaction of neutrons (a major component of cosmic rays) with nitrogen in our atmosphere. The *C-14 then combines with oxygen in the atmosphere to produce *C-14 carbon dioxide, which eventually makes its way into living plants. The *C-14 then enters the food chain and enters the bodies of living animals. The *C-14 in living plants and animals gradually decreases in concentration with time due to radioactive decay, but it is continually replenished by the intake of additional *C-14 through plant respiration and the food chain. Eventually, a constant equilibrium concentration of *C-14 develops in the living plants and animals, in parallel with a corresponding equilibrium concentration of the non-radioactive isotope of carbon, C-12. Both C-12 and *C-14 are regulated to equilibrium through their absorption (as C-12 and *C-14 carbon dioxide) and their subsequent excretion as sugars. The difference here is that the C-12 does not radioactively decay while *C-14 does, so the equilibrium achieved is at a lower plant concentration for *C-14 than for C-12. In animals, carbon is absorbed via plant food and excreted as carbon dioxide in breath. The net result of the above processes is that the ratio [*C-14 / C-12] in living plants and animals also equilibrates to a constant value. When the plant or animal dies, the absorption of C-12 and *C-14 abruptly ceases. Consequently, while the C-12 that was present at the time of death remains at a fixed concentration, the *C-14 that was present at the time of death gradually decreases in concentration through radioactive decay with a half-life of 28,650 years. Therefore, the ratio of [*C-14 / C-12] also decreases with time after death of the plant or animal. After about 5 half-lives of *C-14 (i.e., about 716,250 years), this ratio has fallen to almost zero (5 half-lives is a rule-of-thumb for estimating the time required for a radionuclide to decay to approximately 1% of its initial activity). By accurately measuring the ratio of [*C-14 / C-12] within this time window using sensitive nuclear analytic instruments such as mass spectrometers, the time since death of a living organism can be determined quite accurately over a time scale of approximately 60,000 years. The Influence of Nuclear Weapons Testing on Carbon Dating Carbon dating was more accurate prior to the 1950s than it is today. That is because after the 1950s, above ground testing of nuclear weapons by China, the U.S., and the Soviet Union altered the previously constant [*C-14 / C-12] ratio in our atmosphere, partially invalidating the theory of carbon dating determinations. Although approximate corrections can be made, the ultimate accuracy of carbon dating became significantly lower after the 1950s. Radiometric Dating of Inanimate Materials Using Other Radionuclides Other forms of radiometric dating, relying on the absorption of radionuclides other than C-14 by living organisms or inanimate materials, provides the ability to date animal and plant materials over much longer time scales than is possible with carbon dating, and is used to date the inorganic mineral component in animal bones as well as minerals and rocks. The principle of these other radiometric dating techniques is based on measuring the decay rate of one specific radionuclide relative to an assumed fixed concentration of a second radionuclide in the material of interest that has a much longer half-life. Potassium-argon and uranium-lead dating are the most common examples of this method of radiometric dating. Potassium–argon (abbreviated K–Ar) dating is used most frequently in geochronology and archaeology. This method is based on a very precise measurement of the conversion rate of a radioactive isotope of potassium *K-40 due to radioactive decay into the stable gas Argon-40. Potassium contains a trace quantity of the naturally occurring radionuclide *K-40 and is a common element found in many materials such as micas, clays, and minerals. In these materials, argon-40, the gaseous stable decay product of *K-40, is able to escape these materials when they are in a molten or uncrystalized state, but is prevented from escaping after they have solidified or recrystallized, and therefore starts to accumulate. The time since a rock sample solidified or recrystallized is obtained by accurately measuring the ratio of the Ar-40 accumulated to the amount of *K-40 remaining in the rock. The extremely long half-life of *K-40 (1.26 billion years) allows this method of radiometric dating to be used to calculate the absolute age of samples of rock from a few thousand years to a few billion years, as well as the age of the earth itself. Other radionuclide pairs used in this form of radiometric dating are uranium-lead, rubidium-strontium, and uranium-thorium. The choice of radionuclides depends on the chemical form of the materials to be dated, on the age-range anticipated, and on the accuracy required. Summary Radiometric dating has contributed immeasurably to our understanding of geological history and has contributed greatly to anthropological research. However, the technique requires exquisitely sensitive and accurate nuclear measurement equipment which usually only dedicated laboratories possess. Carbon dating is often used when the age of organic matter needs to be measured over a timescale of about 60,000 years. The accuracy of carbon dating was compromised after the 1950s when the C-14/C-12 ratio in our atmosphere was changed due to nuclear weapon testing by the U.S., Russia, and China. For measuring longer intervals of time in inorganic materials—rocks, minerals, or the earth itself--the decay rates of one radionuclide relative to an assumed fixed concentration of a second radionuclide having a much longer half-life are measured. Potassium-argon and uranium-lead dating are the most common examples of this method of dating. The former is used most frequently in geochronology and archaeology. |
About the Author Archives |
Live Chat Support
×
Connecting

You:
::content::
::agent_name::
::content::
::content::
::content::


 RSS Feed
RSS Feed